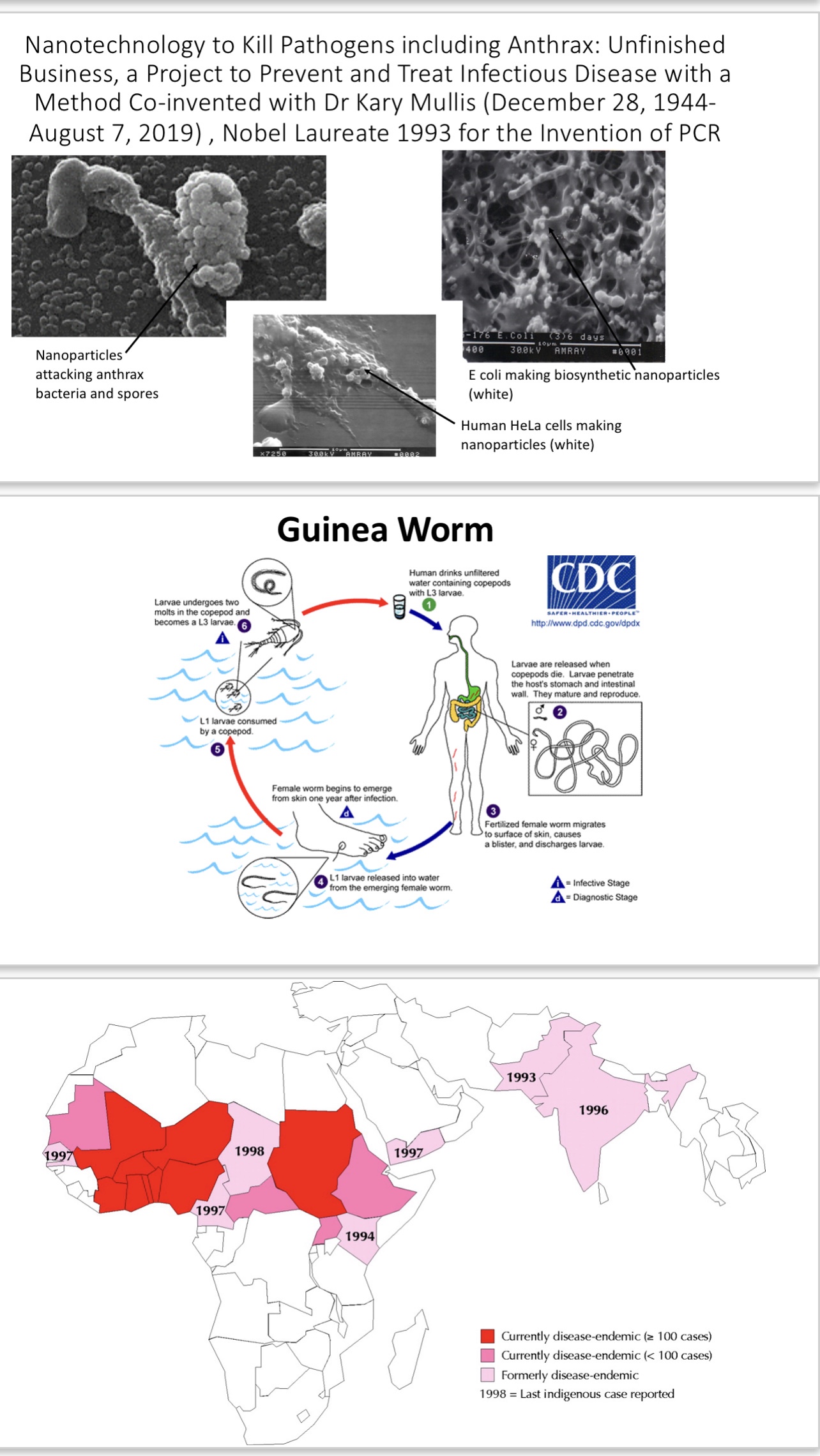
My colleagues and I answered a call for innovative ideas to fund rapidly responding to emerging infectious disease in a timely and effective way, before COVID-19 was a thought, from The Coalition for Epidemic Preparedness Innovations (CEPI), a global partnership launched in 2017 to develop vaccines to stop future epidemics. We were flatly turned down. CARB-X was launched July 28, 2016, by two divisions of the US Department of Health and Human Services (HHS): the Biomedical Advanced Research and Development Authority (BARDA), which is part of the Office of the Assistant Secretary for Preparedness and Response (ASPR) and the National Institute of Allergy and Infectious Diseases. We had sent in a proposal with a well-known institute to CARB-X in 2017, which was not funded. On 7 January 2020, CARB-X released a press release that CARB-X backs Centauri (a UK biotech company) to advance a new platform that combines antibiotic power with the ability to boost the immune system to fight infections caused by drug-resistant bacteria (this was invented by Dr Kary Mullis, Nobel Laureate and further improved by support from the USAF AFRL Brooks Counterproliferation Team) Centauri therapeutics.https://carb-x.org/carb-x-news/carb-x-backs-centauri-to-advance-a-new-platform-that-combines-antibiotic-power-with-the-ability-to-boost-the-immune-system-to-fight-infections-caused-by-drug-resistant-bacteria/ Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X) is a global non-profit partnership for accelerating antibacterial research to address global rising threat of drug-resistant bacteria. It controls up to US$480 million to invest, 2016-22; CARB-X funds the best science from around the world. The CARB-X portfolio is the world’s largest early development pipeline of new antibiotics, vaccines, rapid diagnostics and other products to prevent, diagnose and treat life-threatening bacterial infections. CARB-X is led by Boston University and funded by US Department of Health and Human Services Biomedical Advanced Research and Development Authority (BARDA), part of the Office of the Assistant Secretary for Preparedness and Response (ASPR), the Wellcome Trust, a global charity based in the UK working to improve health globally, Germany’s Federal Ministry of Education and Research (BMBF), the UK Government’s Global Antimicrobial Resistance Innovation Fund (UK GAMRIF), the Bill & Melinda Gates Foundation, the world’s largest foundation dedicated to improving the quality of life for individuals around the world, and receives in-kind support from National Institute of Allergy and Infectious Diseases (NIAID), part of the US National Institutes of Health (NIH). CARB-X headquarters are at Boston University School of Law. BARDA and the Gates Foundation were appealed to in 2013 and 2012, respectively, for support of the US (our proposal, submitted again to CEPI in October 2019, with Dr Mullis’s collaboration just prior to his passing on August 7, 2019) improvements in chemically programmable immunity using the Nanobe platform, but we were totally ignored by these two. How many times can a good idea be turned down? Until it becomes someone else’s.
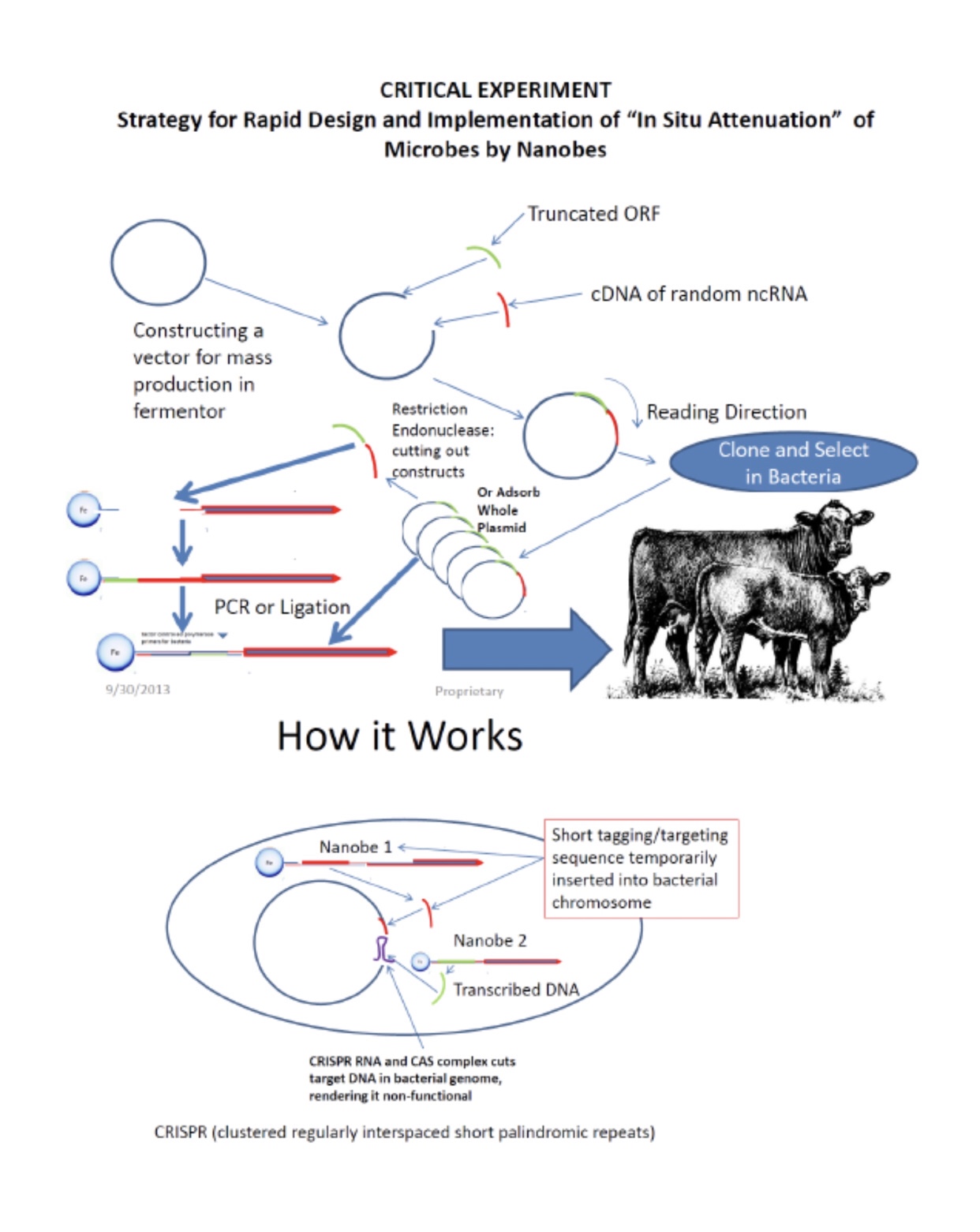
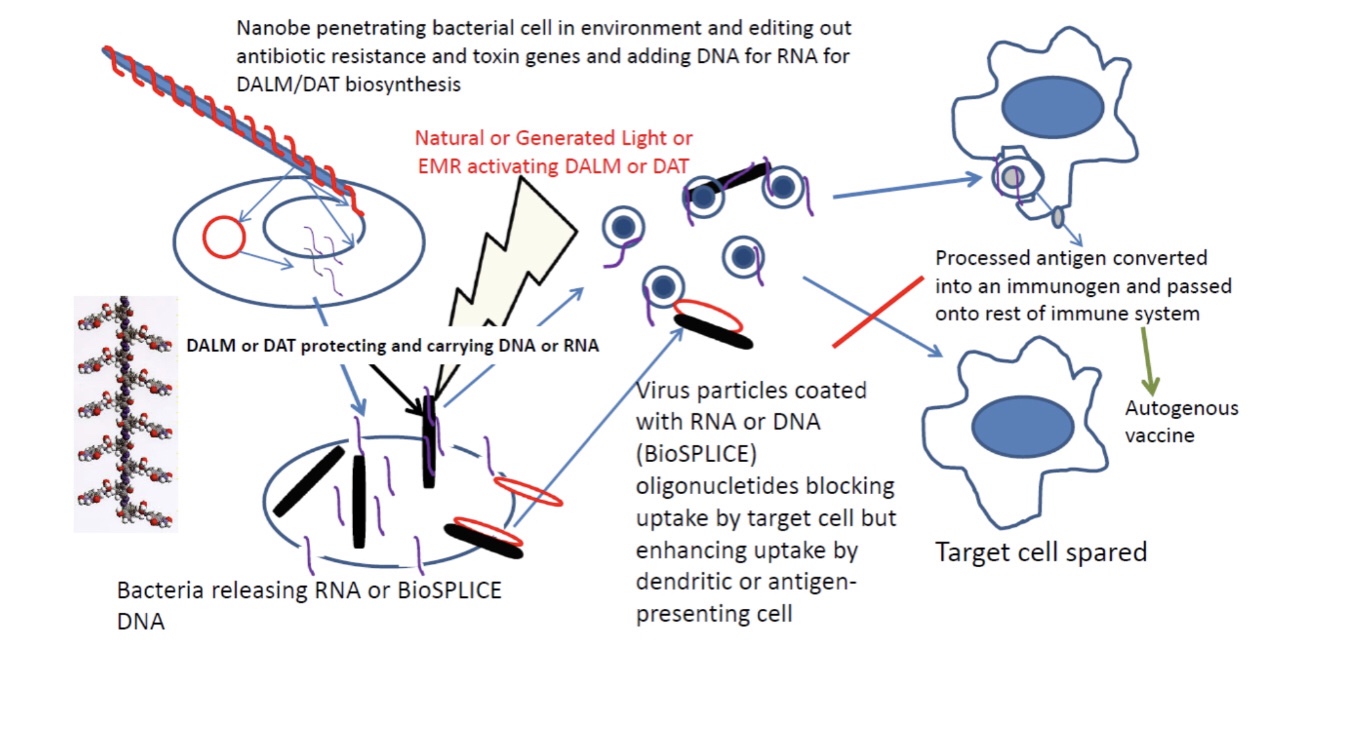
For CEPI:
Target pathogens
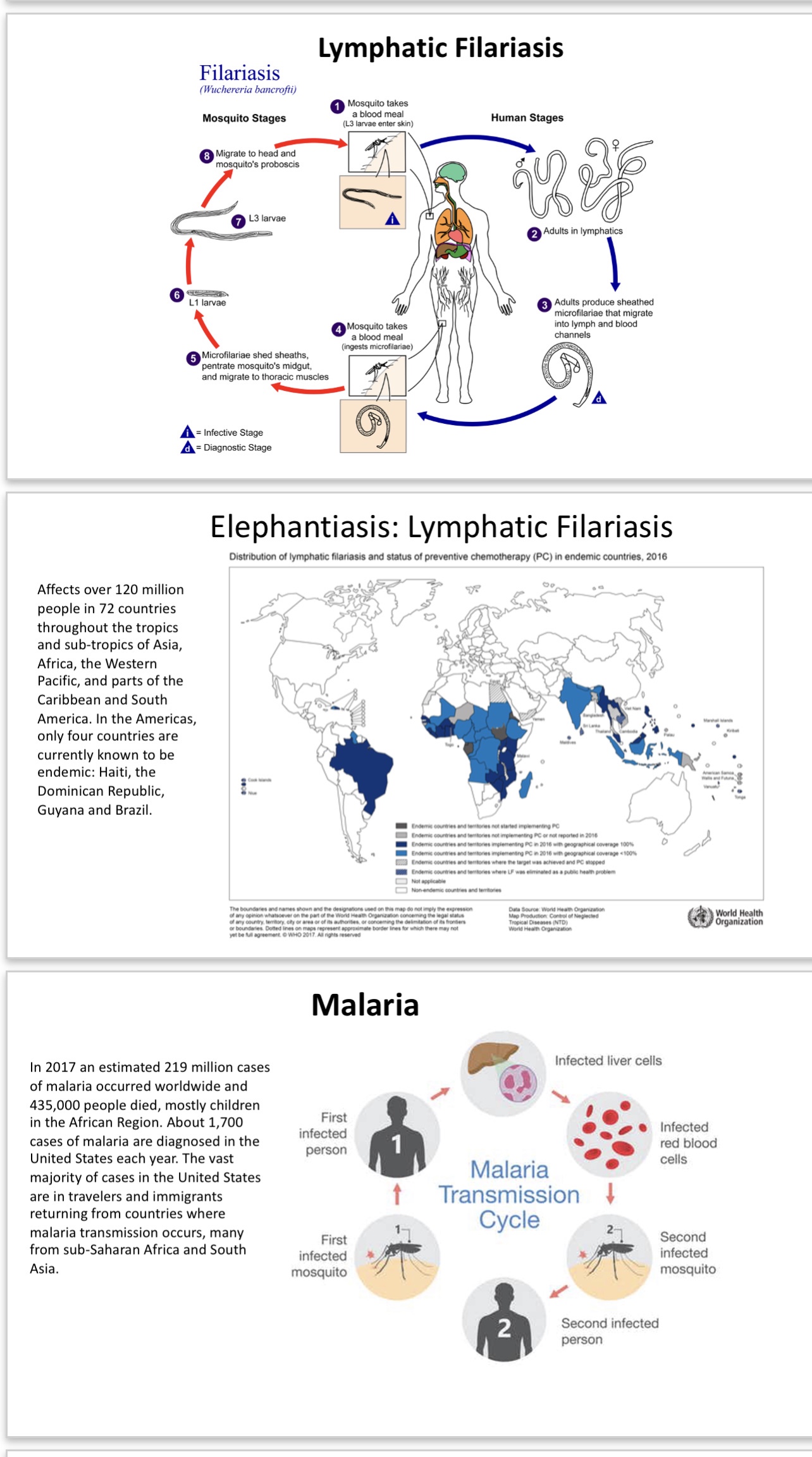
Pathogen 1: Malaria (Plasmodium falciparum and others): There is no current effective vaccine for this disease and resistance to the most effective treatment with artemisinin is spreading. However, natural resistance has been demonstrated in endemic areas in children, but the effective response requires IgM and not IgG and the target antigen galactose-alpha-1,3-galactose. Vaccines that lead to IgG response development will not be as effective as those that direct the immune system toward an IgM response to alpha gal.
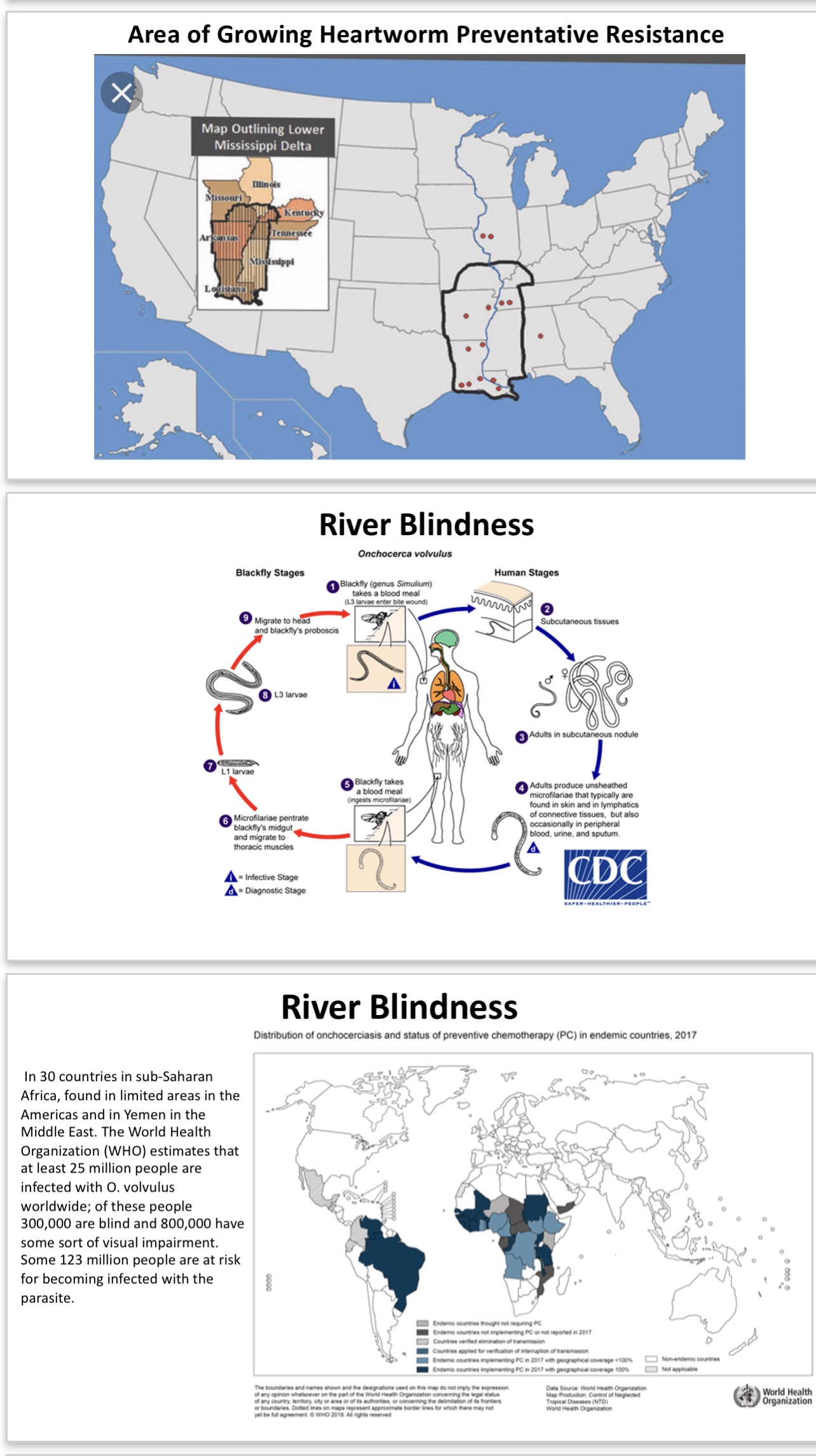
Pathogen 2: Heartworms (Dirofilaria immitis): this disease is not only pervasive in canines across the world, but the current treatment (may be called a “preventative“ but actually kills as a microfilaricide after infection but before worm maturation) uses the same macrocyclide lactones, to which resistance is developing and spreading, used in human filariasis; macrocyclide lactones are the only class of drugs currently available to treat these filarial diseases, which means the development of resistance would eliminate any effective treatment, and they involve altering or blocking the release of an antigen which interferes with the immune response to these filaria and which is effectively activated upon treatment to eliminate microfilaria. There is a robust antibody response which is interfered with by soluble antigen/antibody complex formation which could be overcome by the appropriate redirection or facilitation of immune targeting.
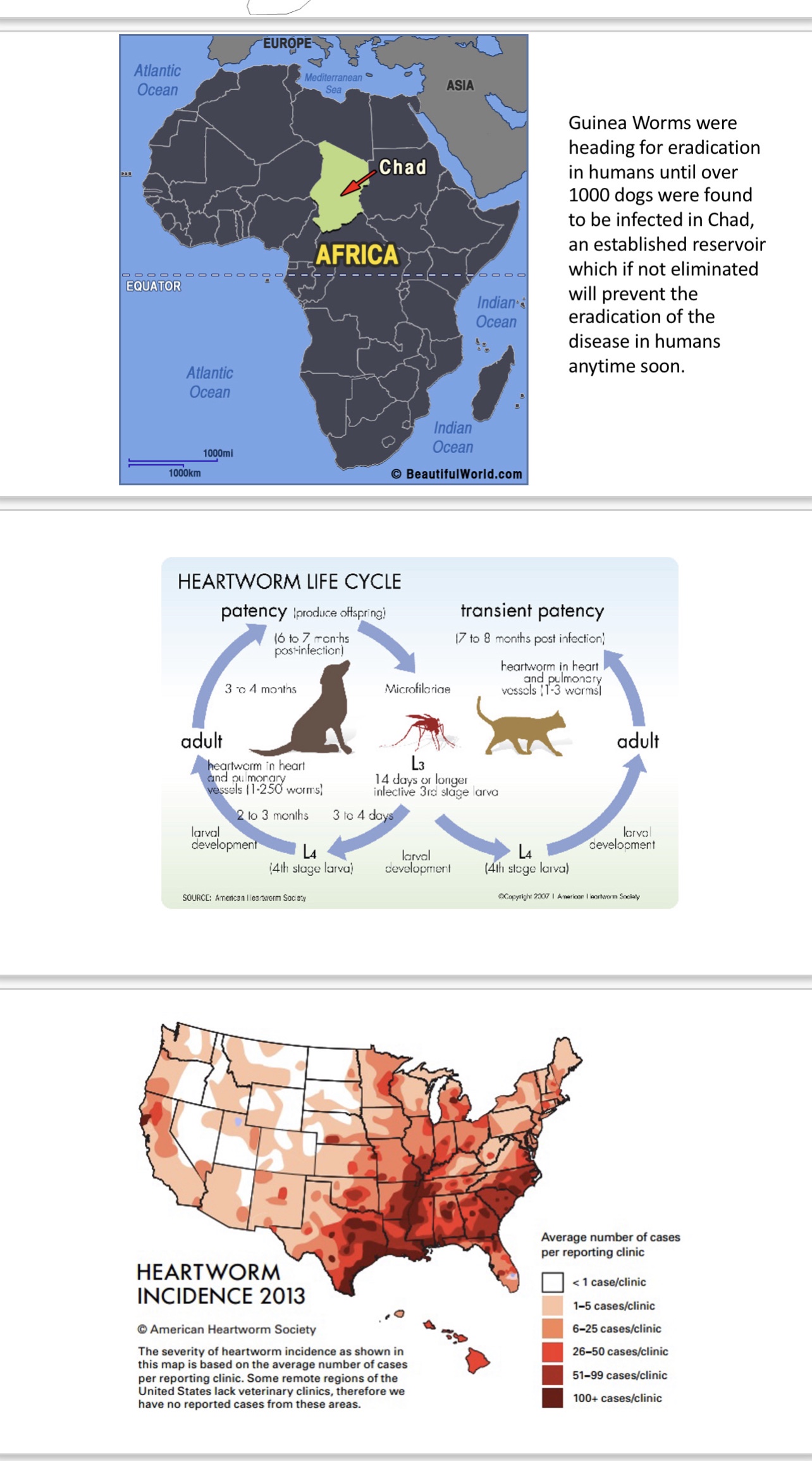
Pathogen 3: Dracunculus medinensis, Guinea worm; the hope to eradicate this parasite, now only in Africa, has recently shown signs of failure after finding that the once thought to be rare infection in dogs (over a thousand infected dogs discovered) is now commonly present in them in Chad. This infection of dogs in areas of re-emerging human infection allows for studies of the platform response in dogs under field conditions which would be most relevant to concurrent human disease.